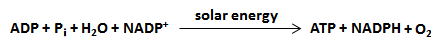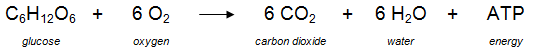Energy and Matter cycles
The Carbon-Oxygen Cycle
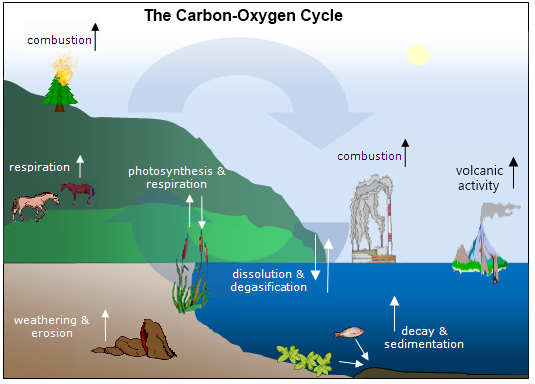
Carbon and oxygen are necessary for all organisms. These elements move between producers, consumers, and the atmosphere in a continuous biogeochemical cycle.The carbon and oxygen cycles are sometimes discussed separately.However, these cycles can also be addressed together since they are dependent upon each other for proper operation. This combined, interdependent cycle is known as the carbon-oxygen cycle.
Processes That Release Carbon Dioxide into the Atmosphere
Processes That Store Carbon Dioxide
Processes That Release Carbon Dioxide into the Atmosphere
- Cellular respiration is a natural process in which substances are broken down to create energy.
- Decay is a natural process in which organic matter, like dead organisms,decomposes or breaks down.
- Volcanic activity is a natural process involving the eruption of hot gases and rock material from within the Earth.
- Weathering is the process by which stones at the Earth's surface are broken down, either physically or chemically.
- Combustion is a reaction in which a substance burns in oxygen.
- It can be natural, such as a forest fire started by lightning.
- It can be man-made, such as burning wood and fossil fuels.
- Degasification is the release of carbon dioxide that was once dissolved in water into the air. It is a natural process, but it has increased due to higher global temperatures. (Warm water can hold less dissolved gases than cooler water. This is why a can of soda goes flat when it gets warm.)
Processes That Store Carbon Dioxide
- Photosynthesis is a natural process in which carbon dioxide and water are converted into sugar.
- Sedimentation is a natural process in which pieces of rock and other matter settles out of water and are buried.
- Dissolution is a natural process in which carbon dioxide from the atmosphere dissolves into water.
Photosynthesis & Respiration
The two most important natural processes that drive the carbon-oxygen cycle are photosynthesis and cellular respiration. Each of these processes must take place in order for the cycle to function properly. This is because the end products of one process are starting materials for the the other.
The black arrows represent the flow of oxygen and carbon dioxide due to cellular respiration. The magenta arrows represent the flow of gases due to photosynthesis.
Photosynthesis:
carbon dioxide + water → sugar + oxygen
The end products of photosynthesis are sugar and oxygen. These substances are the starting materials of cellular respiration.
Cellular Respiration:
sugar + oxygen →carbon dioxide + water
The end products of cellular respiration are carbon dioxide and water. These substances are the starting materials of photosynthesis.
The black arrows represent the flow of oxygen and carbon dioxide due to cellular respiration. The magenta arrows represent the flow of gases due to photosynthesis.
Photosynthesis:
carbon dioxide + water → sugar + oxygen
The end products of photosynthesis are sugar and oxygen. These substances are the starting materials of cellular respiration.
Cellular Respiration:
sugar + oxygen →carbon dioxide + water
The end products of cellular respiration are carbon dioxide and water. These substances are the starting materials of photosynthesis.
Carbon Dioxide in the Atmosphere
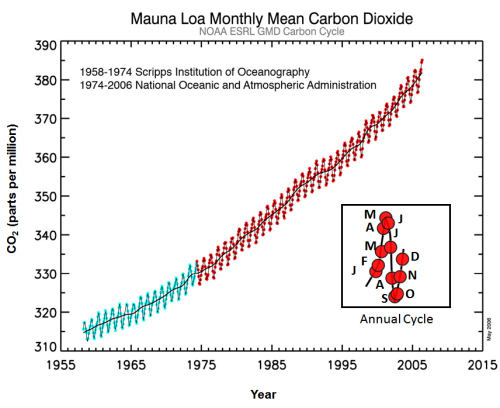
Currently, the overall amount of carbon dioxide in the atmosphere is increasing. This is because carbon dioxide is being released at a faster rate than it is being stored. According to a report from the August 2011 report from NOAA-ESRL, the amount of carbon dioxide has increased almost 25% (from 315 ppm to 392 ppm) since 1958.
The blue portion of the graph represents the data collected by Scripps. The data collected by NOAA-ESRL is represented in red. The mid-line represents the average CO2 concentrations for the year.
Adapted from figure courtesy of NOAA.
The graph to the left shows the monthly concentrations of carbon dioxide in the atmosphere. These measurements are taken at an observatory in Mauna Loa, Hawaii. The location of this observatory is important because it affects the data. Mauna Loa is far from cities (where carbon dioxide levels are higher). It is also located in a part of the world where the air is well-mixed. So, these measurements provide a good representative for the entire planet.
Though the overall concentration is increasing, there are also fluctuations in the levels of carbon dioxide in the atmosphere throughout the year. This is due to seasonal changes.
Since most plants are located in the northern hemisphere, the total amount of photosynthesis slows down during the winter months.Therefore, plants are removing less carbon dioxide from the atmosphere.When photosynthesis increases again during the spring and summer, the CO2 levels drop again.
The blue portion of the graph represents the data collected by Scripps. The data collected by NOAA-ESRL is represented in red. The mid-line represents the average CO2 concentrations for the year.
Adapted from figure courtesy of NOAA.
The graph to the left shows the monthly concentrations of carbon dioxide in the atmosphere. These measurements are taken at an observatory in Mauna Loa, Hawaii. The location of this observatory is important because it affects the data. Mauna Loa is far from cities (where carbon dioxide levels are higher). It is also located in a part of the world where the air is well-mixed. So, these measurements provide a good representative for the entire planet.
Though the overall concentration is increasing, there are also fluctuations in the levels of carbon dioxide in the atmosphere throughout the year. This is due to seasonal changes.
Since most plants are located in the northern hemisphere, the total amount of photosynthesis slows down during the winter months.Therefore, plants are removing less carbon dioxide from the atmosphere.When photosynthesis increases again during the spring and summer, the CO2 levels drop again.
The Nitrogen Cycle
The nitrogen cycle is the cycle of consumption and regeneration of nitrogen within our environment.
Overview

Nitrogen is an essential component of amino acids (proteins) and nucleic acids (DNA and RNA). Therefore, all organisms require nitrogen to survive. Even though nitrogen is the most abundant gas in the atmosphere, most organisms are unable to use this form of nitrogen.However, there are a few microscopic organisms and natural processes, such as lightening, that can convert unusable nitrogen in the atmosphere to usable forms of nitrogen.
During the nitrogen cycle, atmospheric nitrogen (N2) is fixed or changed into nitrogen containing compounds, such as ammonia or nitrates, by nitrogen-fixing bacteria. Plants can then absorb the nitrogen compounds from the soil and use it to form chlorophyll and other important biological molecules.
Consumers must obtain nitrogen from the organisms they consume.Herbivores receive their nitrogen from the plants that they eat, and carnivores get their nitrogen from the animals they consume. However, all organisms depend on the ability of nitrogen-fixing microbes to convert atmospheric nitrogen into a form of nitrogen that plants can assimilate, or take in and use.
Finally, nitrogen is returned to the atmosphere through the combustion of fossil fuels or when bacteria or fungi break down the nitrogen found in fertilizers, urine, and dead plants and animals.
During the nitrogen cycle, atmospheric nitrogen (N2) is fixed or changed into nitrogen containing compounds, such as ammonia or nitrates, by nitrogen-fixing bacteria. Plants can then absorb the nitrogen compounds from the soil and use it to form chlorophyll and other important biological molecules.
Consumers must obtain nitrogen from the organisms they consume.Herbivores receive their nitrogen from the plants that they eat, and carnivores get their nitrogen from the animals they consume. However, all organisms depend on the ability of nitrogen-fixing microbes to convert atmospheric nitrogen into a form of nitrogen that plants can assimilate, or take in and use.
Finally, nitrogen is returned to the atmosphere through the combustion of fossil fuels or when bacteria or fungi break down the nitrogen found in fertilizers, urine, and dead plants and animals.
Phosphorus Cycle

he phosphorus cycle is the cycle of consumption and regeneration of phosphorus within our environment.Phosphorus is the second most abundant mineral in the human body since it is a component of many important compounds, including DNA, RNA, and ATP.
Phosphorus is mainly derived from the weathering of phosphate rocks, although it can also come from phosphate fertilizers, the decomposition of dead plants and animals, and the excreta (fecal matter) of animals. The atmosphere or air is not a source of phosphorus, however, because at normal temperatures and pressures, phosphorus and phosphoric compounds occur as solids, not as gases. This is different from other biogeochemical cycles, such as the nitrogen cycle or the water cycle, where at least one portion of the cycle passes through a gaseous phase.
Phosphorus is mainly derived from the weathering of phosphate rocks, although it can also come from phosphate fertilizers, the decomposition of dead plants and animals, and the excreta (fecal matter) of animals. The atmosphere or air is not a source of phosphorus, however, because at normal temperatures and pressures, phosphorus and phosphoric compounds occur as solids, not as gases. This is different from other biogeochemical cycles, such as the nitrogen cycle or the water cycle, where at least one portion of the cycle passes through a gaseous phase.
The Water Cycle
Water and energy are transferred throughout the hydrosphere, lithosphere and atmosphere during the water cycle, or the hydrologic cycle.
The amount of water on Earth remains constant, but it continuously changes forms as energy from the Sun drives the cycle.
The amount of water on Earth remains constant, but it continuously changes forms as energy from the Sun drives the cycle.
Movement of Water During the Water Cycle

The water cycle describes the continuous movement of water on, above, and below the surface of the Earth. Actually, there is much more water being stored in the cycle than is moving through the cycle. Water may be stored for a short time as water vapor in the atmosphere, for days or weeks in a lake, or for thousands of years in a polar ice cap.
The water cycle is a cycle with no beginning or end. It includes the following processes:
Condensation is the changing of gas to a liquid (water vapor to water) and is crucial for the formation of clouds. Clouds form in the atmosphere when air containing water vapor rises and cools. Water vapor can be present in the air even when clouds are not visible. Clouds become visible when water particles combine with each other around tiny particles of dust to form water droplets.
Water returns to the Earth as precipitation. Precipitation is the process by which water vapor in the air condenses to form drops heavy enough to fall to the Earth's surface. During infiltration water fills the porous spaces in the lithosphere. Surface runoff occurs when no more water can be absorbed into the ground and gravity pulls it downhill. Water flows over land and forms rivers. Rivers usually flow into the ocean, and the water cycle continues from there.
Evaporation and transpiration are similar in that they are both processes in which water is changed into water vapor. Evaporation often happens as a result of heat – liquid water is heated until it turns to a gas (water vapor) and is released into the atmosphere. Transpiration is the process by which water is carried through plants, from roots to leaves, where it changes to water vapor and is released to the atmosphere.
Sublimation is the changing of water from a solid directly to a gas with no intermediate liquid stage. The opposite of sublimation is deposition, when water vapor changes directly to a solid. Snowflakes and frost are examples of deposition.
The water cycle is a cycle with no beginning or end. It includes the following processes:
Condensation is the changing of gas to a liquid (water vapor to water) and is crucial for the formation of clouds. Clouds form in the atmosphere when air containing water vapor rises and cools. Water vapor can be present in the air even when clouds are not visible. Clouds become visible when water particles combine with each other around tiny particles of dust to form water droplets.
Water returns to the Earth as precipitation. Precipitation is the process by which water vapor in the air condenses to form drops heavy enough to fall to the Earth's surface. During infiltration water fills the porous spaces in the lithosphere. Surface runoff occurs when no more water can be absorbed into the ground and gravity pulls it downhill. Water flows over land and forms rivers. Rivers usually flow into the ocean, and the water cycle continues from there.
Evaporation and transpiration are similar in that they are both processes in which water is changed into water vapor. Evaporation often happens as a result of heat – liquid water is heated until it turns to a gas (water vapor) and is released into the atmosphere. Transpiration is the process by which water is carried through plants, from roots to leaves, where it changes to water vapor and is released to the atmosphere.
Sublimation is the changing of water from a solid directly to a gas with no intermediate liquid stage. The opposite of sublimation is deposition, when water vapor changes directly to a solid. Snowflakes and frost are examples of deposition.
Biological Organic Molecules
A group of atoms that is held together by covalent bonds is known as a molecule. When bonding occurs between two or more carbon atoms, the group is known as an organic molecule.
Biological Organic Molecules
Biological molecules are composed of small repeating subunits that bond together to form larger units. The subunits, or building blocks, are calledmonomers. Polymers are the complex molecules formed from the repeating subunits.
There are four basic classes of complex biological organic molecules, ormacromolecules, that compose cells: carbohydrates, proteins, lipids, and nucleic acids. Each of the major classes of biological molecules is associated with different properties and functions within cells and whole organisms.
There are four basic classes of complex biological organic molecules, ormacromolecules, that compose cells: carbohydrates, proteins, lipids, and nucleic acids. Each of the major classes of biological molecules is associated with different properties and functions within cells and whole organisms.
Carbohydrates
Carbohydrates are organic macromolecules that are made up of carbon, hydrogen, and oxygen atoms. These atoms are combined in a ratio of:
1 carbon atom : 2 hydrogen atoms : 1 oxygen atom
The presence of multiple carbon-hydrogen bonds within carbohydrates makes them an excellent source of energy. (The energy is released when these bonds are broken.)
Carbohydrates may be simple or complex. The building blocks of carbohydrates are the simple sugars known as monosaccharides.Sugars such as glucose, fructose, and ribose are all examples of monosaccharides.
Monosaccharides can be combined to form more complex carbohydrates known as polysaccharides. Glycogen, starch, and cellulose are all examples of polysaccharides. These compounds are typically used for long term energy storage or as structural molecules. Cellulose, for example, is a major component found in the cell walls of plants.
Dietary fiber is a special class of carbohydrates that cannot be digested by the human body. Cellulose is one example of a carbohydrate that acts as fiber. Dietary fiber is an important part of a healthy diet because it is essential for proper digestion. Humans can get fiber by eating many different kinds of plants, such as whole grains, legumes, prunes, and potatoes.
Carbohydrates are organic macromolecules that are made up of carbon, hydrogen, and oxygen atoms. These atoms are combined in a ratio of:
1 carbon atom : 2 hydrogen atoms : 1 oxygen atom
The presence of multiple carbon-hydrogen bonds within carbohydrates makes them an excellent source of energy. (The energy is released when these bonds are broken.)
Carbohydrates may be simple or complex. The building blocks of carbohydrates are the simple sugars known as monosaccharides.Sugars such as glucose, fructose, and ribose are all examples of monosaccharides.
Monosaccharides can be combined to form more complex carbohydrates known as polysaccharides. Glycogen, starch, and cellulose are all examples of polysaccharides. These compounds are typically used for long term energy storage or as structural molecules. Cellulose, for example, is a major component found in the cell walls of plants.
Dietary fiber is a special class of carbohydrates that cannot be digested by the human body. Cellulose is one example of a carbohydrate that acts as fiber. Dietary fiber is an important part of a healthy diet because it is essential for proper digestion. Humans can get fiber by eating many different kinds of plants, such as whole grains, legumes, prunes, and potatoes.
Lipids
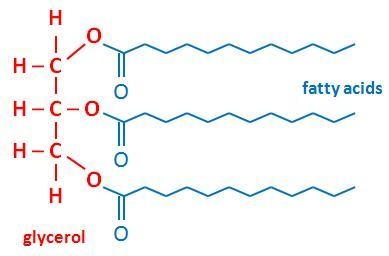
Lipids are organic macromolecules that are insoluble in water. This is why lipids are often found in biological membranes and other waterproof coverings (e.g. plasma membrane, intracellular membranes of organelles).These lipids play a vital role in regulating which substances can or cannot enter the cell.
The most important lipids, however, are fats. Triglycerides are a type of fat that contain one glycerol molecule and three fatty acids.
Fatty acids are long chains of CH2 units joined together. The fatty acids insaturated fats do not contain any double bonds between the CH2 units whereas the fatty acids in unsaturated fats contain some carbon-carbon double bonds. Saturated fats are found in butter, cheese, chocolate, beef, and coconut oil. Unsaturated fats are found in olives and olive oil, peanuts and peanut oil, fish, and mayonnaise.
Fats are important because they are a major source of energy. Since they contain even more carbon-hydrogen bonds than carbohydrates, fatty tissue has the ability to store energy for extended periods of time
The most important lipids, however, are fats. Triglycerides are a type of fat that contain one glycerol molecule and three fatty acids.
Fatty acids are long chains of CH2 units joined together. The fatty acids insaturated fats do not contain any double bonds between the CH2 units whereas the fatty acids in unsaturated fats contain some carbon-carbon double bonds. Saturated fats are found in butter, cheese, chocolate, beef, and coconut oil. Unsaturated fats are found in olives and olive oil, peanuts and peanut oil, fish, and mayonnaise.
Fats are important because they are a major source of energy. Since they contain even more carbon-hydrogen bonds than carbohydrates, fatty tissue has the ability to store energy for extended periods of time
Proteins
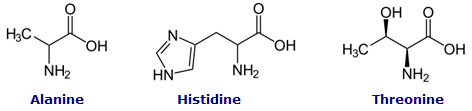
Proteins are organic macromolecules that are composed of amino acidmonomers. There are 20 essential amino acids that are used by all living things to construct proteins. These amino acids are made up of the elements carbon, hydrogen, oxygen, and nitrogen. Some of the amino acids also contain sulfur. Three of the amino acids are shown below.
Proteins differ from each other due to the number and arrangement of their component amino acids. Proteins also take on unique shapes as determined by their amino acid sequences.
Water is the most abundant molecule in the body, but proteins are the second most abundant type of molecule. Proteins assist with muscular contractions and serve many structural roles. For example, cartilage and tendons are made of a protein known as collagen, and a protein known as keratin is found in hair, nails, feathers, hooves, and some animal shells.
Proteins are also involved in cell signaling, cell transport, immune responses, and the cell cycle. Hormones are protein-containing substances that play a role in the regulation of cellular functions.Substances containing proteins that play a role in defense are antibodies.
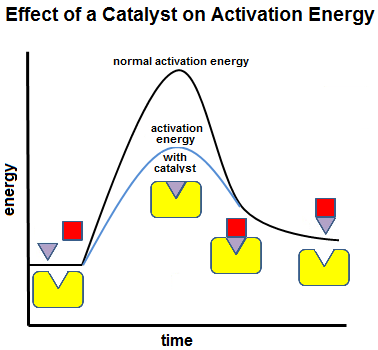
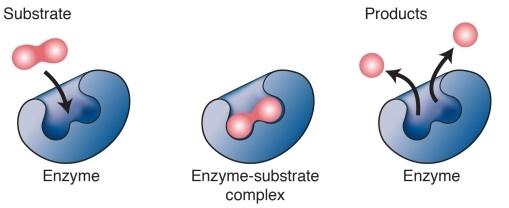
Other proteins known as enzymes are involved in many biological processes such as the breakdown of food molecules, transport across the cell membrane, and cellular signaling and regulation. Most enzymes have a surface with one or more deep folds. The folds make pockets calledactive sites. The active sites match folds in the substrate's surface, like a key matches a lock. So, the enzyme only catalyzes specific reactions.Once an enzyme binds to a substrate, the amount of energy needed to start a chemical reaction with the substrate is reduced. In some cases, the activation energy is too high to overcome without an enzyme.
Proteins differ from each other due to the number and arrangement of their component amino acids. Proteins also take on unique shapes as determined by their amino acid sequences.
Water is the most abundant molecule in the body, but proteins are the second most abundant type of molecule. Proteins assist with muscular contractions and serve many structural roles. For example, cartilage and tendons are made of a protein known as collagen, and a protein known as keratin is found in hair, nails, feathers, hooves, and some animal shells.
Proteins are also involved in cell signaling, cell transport, immune responses, and the cell cycle. Hormones are protein-containing substances that play a role in the regulation of cellular functions.Substances containing proteins that play a role in defense are antibodies.
Other proteins known as enzymes are involved in many biological processes such as the breakdown of food molecules, transport across the cell membrane, and cellular signaling and regulation. Most enzymes have a surface with one or more deep folds. The folds make pockets calledactive sites. The active sites match folds in the substrate's surface, like a key matches a lock. So, the enzyme only catalyzes specific reactions.Once an enzyme binds to a substrate, the amount of energy needed to start a chemical reaction with the substrate is reduced. In some cases, the activation energy is too high to overcome without an enzyme.
Nucleic Acids
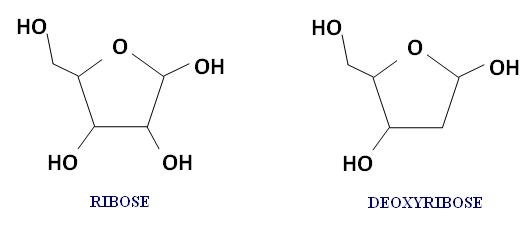
Nucleic acids are formed from nucleotide monomers. Nucleotides are chemical compounds that are primarily comprised of the elements carbon, hydrogen, oxygen, nitrogen, and phosphorus. They consist of a five-carbon sugar, a nitrogenous base, and one or more phosphate groups.
There are two main types of nucleic acids - ribonucleic acids (RNA) anddeoxyribonucleic acids (DNA). These nucleic acids are different because their five-carbon sugars are different. RNA contains ribose, and DNA contains deoxyribose.
DNA and RNA also have different functions. DNA stores genetic information and encodes the sequences of all the cell's proteins. RNA is involved in the direct production of the proteins.
Nucleic acids are also different because the sequence of nitrogenous bases that they contain are different. There are five nitrogenous bases found in nucleic acids. Adenine (A), cytosine (C), and guanine (G) are found in both DNA and RNA. Thymine (T) is only found in DNA, anduracil (U) is only found in RNA.
There are two main types of nucleic acids - ribonucleic acids (RNA) anddeoxyribonucleic acids (DNA). These nucleic acids are different because their five-carbon sugars are different. RNA contains ribose, and DNA contains deoxyribose.
DNA and RNA also have different functions. DNA stores genetic information and encodes the sequences of all the cell's proteins. RNA is involved in the direct production of the proteins.
Nucleic acids are also different because the sequence of nitrogenous bases that they contain are different. There are five nitrogenous bases found in nucleic acids. Adenine (A), cytosine (C), and guanine (G) are found in both DNA and RNA. Thymine (T) is only found in DNA, anduracil (U) is only found in RNA.
Enzymes
Enzymes are biological catalysts that lower the activation energy of chemical reactions.
Biological Catalysts
|
Catalysts are substances that speed up chemical reactions by lowering the energy needed to start the reaction. Enzymes are biological catalysts.
The enzyme above lowers the activation energy needed to split the substrate molecule. After the reaction, the enzyme is free to catalyze the breakdown of another reactant molecule.
|
Substances which lower the amount of energy needed to activate a chemical reaction, without being consumed in the reaction, are called catalysts. Enzymes are biological catalysts, generally composed of proteins. By lowering the activation energy, chemical reactions generally occur more rapidly.
Most enzymes are proteins. Like other proteins, enzymes are produced by a cell's ribosomes. Ribosomes produce specific enzymes to act on specific substances, called substrates. For example, the enzyme catalase assists in the breakdown of hydrogen peroxide into water and oxygen. In this case, hydrogen peroxide is catalase's substrate. Many of the chemical reactions that occur in cells are catalyzed by enzymes. The activation energy for many reactions is simply too high to overcome without enzymes, and the reaction will not occur at all in the absence of an enzyme. Without enzymes catalyzing metabolic reactions, cells would not be able to perform metabolism quickly enough to support life. Since enzymes are not consumed in a chemical reaction, their concentration will remain constant unless the cell triggers for re-uptake of the enzymes. Cells can control chemical reactions by producing or removing enzymes. Reaction rates can be increased by increasing the production of enzymes in environments highly concentrated with substrate. Enzymes are also important for the synthesis of new molecules. For example, RNA polymerase is an enzyme that is essential to the process of transcription. Molecules of mRNA are transcribed by RNA polymerases and later new protein molecules are synthesized based on the instructions coded in the mRNA. |
Enzyme Shape & Function
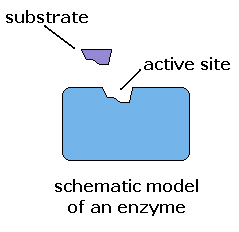
The shape of an enzyme determines how it works. Most enzymes have a surface with one or more deep folds. The folds make pockets, which are called active sites. The active sites match folds in the substrate's surface.Thus, a particular enzyme fits against its substrate like two adjacent puzzle pieces.
An enzyme's shape is key to how the enzyme functions.If its shape is changed, the enzyme may not function as well or at all.Changes in temperature and pH can affect the shape of an enzyme's active sites. Therefore, enzymes are only able to work properly in a certain temperature and pH range.
An enzyme's shape is key to how the enzyme functions.If its shape is changed, the enzyme may not function as well or at all.Changes in temperature and pH can affect the shape of an enzyme's active sites. Therefore, enzymes are only able to work properly in a certain temperature and pH range.
The Interdependence of
Photosynthesis & Respiration
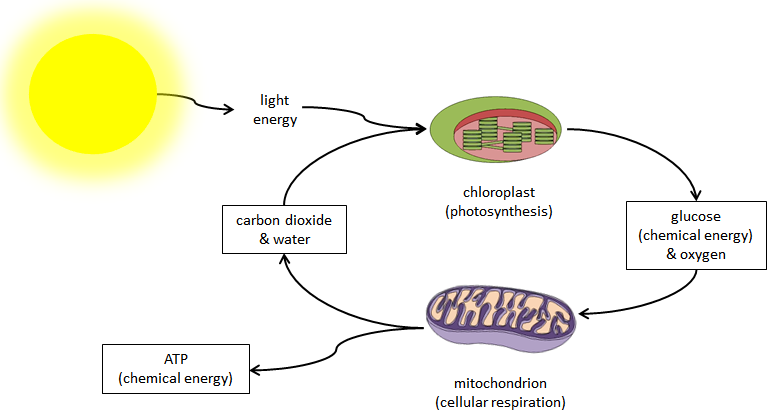
Photosynthesis and cell respiration are complementary processes for cycling carbon dioxide and oxygen as well as transferring energy in ecosystems.Energy moves through ecosystems by the processes of photosynthesis and respiration. During photosynthesis, plants and phytoplankton capture light energy from the Sun and use it to build sugars (chemical energy) out of carbon dioxide and water. The process of photosynthesis can be generally expressed by the equation:
carbon dioxide + water + light sugar + oxygen
Energy from the Sun is essentially stored in the chemical bonds of the sugar molecules in plants. Whenever organisms, including plants, need energy, cells perform cellular respiration. During this process, cells take in oxygen in order to break the bonds of the plant sugars and produce ATP, water, and carbon dioxide.
Cellular energy is stored in the in the phosphate bonds of ATP molecules.Each time a phosphate group is removed from a molecule of ATP, energy is released. This energy can then be used to perform cellular work. The process of cellular respiration can be generally expressed by the equation:
sugar + oxygen carbon dioxide + water + ATP
carbon dioxide + water + light sugar + oxygen
Energy from the Sun is essentially stored in the chemical bonds of the sugar molecules in plants. Whenever organisms, including plants, need energy, cells perform cellular respiration. During this process, cells take in oxygen in order to break the bonds of the plant sugars and produce ATP, water, and carbon dioxide.
Cellular energy is stored in the in the phosphate bonds of ATP molecules.Each time a phosphate group is removed from a molecule of ATP, energy is released. This energy can then be used to perform cellular work. The process of cellular respiration can be generally expressed by the equation:
sugar + oxygen carbon dioxide + water + ATP
ATP
Adenosine triphosphate, or ATP, is the most important biological molecule that supplies energy to cells.
Components of ATP
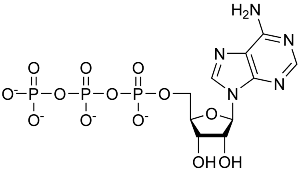
Each molecule of ATP is composed of
- one nitrogenous base (adenine)
- one sugar (ribose)
- three phosphate groups
The ATP-ADP Cycle
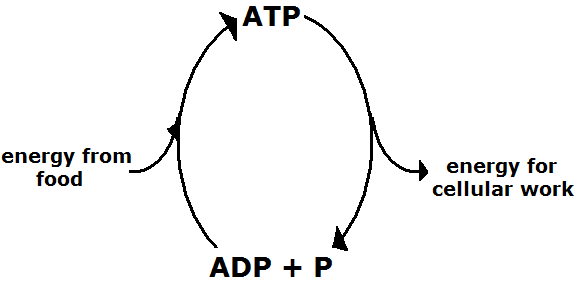
Cells acquire energy from food. This energy is stored as ATP. Cells also use energy for cellular work. When energy is used, a phosphate bond is broken, and ADP (adenosine diphosphate) is formed.
When energy is needed by the cell to perform cellular work, a phosphate bond is broken, and ADP and a free phosphate group are formed.
ATP → ADP + P + energy
When a cell acquires excess energy from food, a phosphate group is reattached to an ADP molecule, and ATP is formed.
ADP + P + energy → ATP
The amount of energy required to attach a phosphate group to ADP is significantly less than the amount of energy that is released when a phosphate bond in ATP is broken.
When energy is needed by the cell to perform cellular work, a phosphate bond is broken, and ADP and a free phosphate group are formed.
ATP → ADP + P + energy
When a cell acquires excess energy from food, a phosphate group is reattached to an ADP molecule, and ATP is formed.
ADP + P + energy → ATP
The amount of energy required to attach a phosphate group to ADP is significantly less than the amount of energy that is released when a phosphate bond in ATP is broken.
Photosynthesis
The ultimate source of energy for most life on Earth is the Sun. Photosynthesis is the overall process by which solar energy chemically converts water and carbon dioxide into chemical energy stored in simple sugars.
The Importance of Photosynthesis
Without photosynthesis, most life on Earth would not exist. This vital process enables producers to transform solar energy into chemical energy. The chemical energy is stored in the bonds of simple sugars until it is needed.
Cells need a constant supply of energy in order to carry out life processes.So, the ability to store and use energy is critical for all life on Earth.
Consumers are unable to transform sunlight into usable energy. Unlike producers, they cannot synthesize complex organic molecules from inorganic matter. Instead, they must consume other organisms that contain the complex molecules and their building blocks. Therefore, producers form the foundation of every food chain on Earth.
Cells need a constant supply of energy in order to carry out life processes.So, the ability to store and use energy is critical for all life on Earth.
Consumers are unable to transform sunlight into usable energy. Unlike producers, they cannot synthesize complex organic molecules from inorganic matter. Instead, they must consume other organisms that contain the complex molecules and their building blocks. Therefore, producers form the foundation of every food chain on Earth.
The Overall Reaction
|
In general, six carbon dioxide molecules and six water molecules are needed to produce one glucose (sugar) molecule and six oxygen molecules.
|
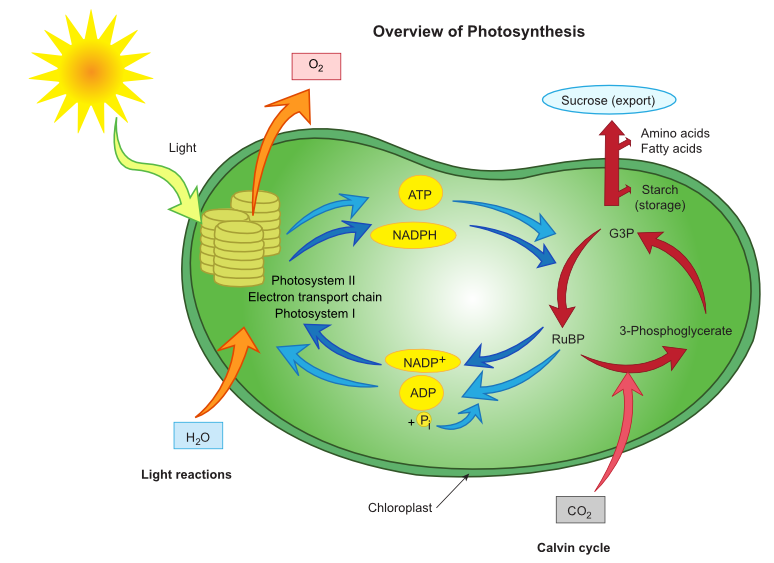
In eukaryotic organisms, such as plants or algae, photosynthesis occurs in the chloroplasts. The overall process of photosynthesis is summarized by the following balanced chemical equation:
In reality, photosynthesis consists of a series of reactions. The equation above shows the initial reactants and final products, but it does not provide the details of every step that occurs during photosynthesis. Photosynthetic reactions are often described as occurring in two separate stages. During the first stage, light energy is used to split water molecules, and oxygen gas is produced as a waste product. Glucose is synthesized from carbon dioxide during the second stage. |
The Light-Dependent Reactions
|
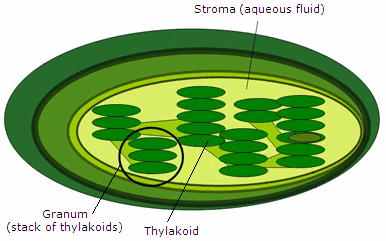
Light-dependent reactions take place in the grana of the thylakoids. Light-independent reaction occur in the fluid-filled space called the stroma.
Leaf surfaces have many tiny openings, called stomata, that are involved in gas exchange. These openings are bordered by guard cells, which regulate the exchange by opening or closing.
|
The first stage of photosynthesis includes the light-dependent reactions.They are called "light-dependent" because they require solar energy.Light-dependent reactions occur within the grana of chloroplasts, such as the one pictured to the left.
Pigment molecules, such as chlorophyll, inside the thylakoids of chloroplasts absorb light energy from the Sun. This causes the electrons inside the chlorophyll molecules to gain energy and move to an excited state. Then, the electrons move from one molecule to another inside the thylakoids, and energy is released with each move. The molecules along which the electrons are passed are together called the electron transport chain. When electrons leave the chlorophyll, water molecules are split into hydrogen and oxygen atoms. The oxygen atoms combine with each other to form oxygen gas (O2), which is released as a waste product through pores (i.e., holes) in the leaves and stems of plants. These pores, also known as stomata, allow the oxygen waste to leave the plant system while also allowing carbon dioxide to enter. Meanwhile, some of the newly formed free hydrogen atoms from the split water molecules donate their electrons to replace the lost chlorophyll electrons. Once the electrons are donated, the remaining protons (i.e., hydrogen ions) move into the stroma of the chloroplast. The other hydrogen atoms, and their electrons and protons, form the coenzyme NADPH, which also moves into the stroma of the chloroplast. A general equation for the light-dependent reactions is summarized to the left. Some energy is also stored when a phosphate (Pi) is added to a molecule of ADP to form ATP. The NADPH's, the protons, and the ATP's made in the light-dependent reactions are later used to synthesize glucose during the dark reactions of photosynthesis. Note that no sugars are synthesized during the light-dependent reactions. |
The Light-Independent Reactions
The second stage of photosynthesis includes the light-independent reactions,otherwise known as the Calvin cycle. These reactions are also known as the dark reactions because they do not require solar energy.However, the term light-independent is more accurate because these reactions can occur in the presence or absence of light.
During the light-independent reactions, the energy stored in ATP and NADPH is released when the molecules are split. This energy is used to produce simple sugars (such as glucose) from carbon dioxide. Simple sugars are used to store chemical energy for use by the cells at later times.
The remaining ADP and NADP+are then available to be recycled during the light-dependent reactions. This process can continue as long as they are exposed to an adequate supply of carbon dioxide, ATP, and NADPH.
Glucose can be used as an energy source through the process of cellular respiration, or it can be converted to organic molecules (such as proteins, carbohydrates, fats/lipids, or cellulose) by various biologic processes.
During the light-independent reactions, the energy stored in ATP and NADPH is released when the molecules are split. This energy is used to produce simple sugars (such as glucose) from carbon dioxide. Simple sugars are used to store chemical energy for use by the cells at later times.
The remaining ADP and NADP+are then available to be recycled during the light-dependent reactions. This process can continue as long as they are exposed to an adequate supply of carbon dioxide, ATP, and NADPH.
Glucose can be used as an energy source through the process of cellular respiration, or it can be converted to organic molecules (such as proteins, carbohydrates, fats/lipids, or cellulose) by various biologic processes.
Summary of the Reactions
The following diagram summarizes the overall process.
During photosynthesis, chloroplasts use solar energy to split water molecules. Oxygen gas is released, and the energy generated from splitting the molecules is used to combine the freed hydrogen ions with carbon dioxide molecules to make glucose molecules.
Limiting Factors
|
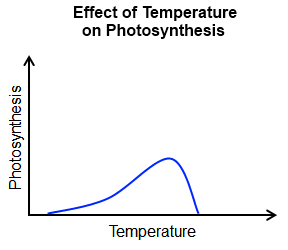
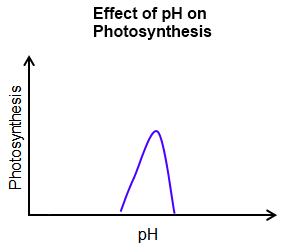
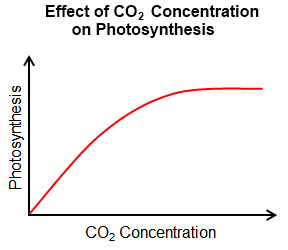
The major factors affecting the rate of photosynthesis include:
Since carbon dioxide and water are the reactants, both must be present for photosynthesis to occur. A decline in the amount of either will slow down the process of photosynthesis. The rate of photosynthesis generally increases as the concentration of carbon dioxide increases. However, the rate stalls once the optimal concentration is reached. The same is true for light intensity. |
Cellular Respiration
The ultimate goal of cellular respiration is to convert the chemical energy in nutrients to chemical energy stored in ATP. Then, the ATP molecules can be used for various cellular metabolic processes.
Cellular Respiration
|
Cellular respiration can take place in the presence or absence of oxygen. However, the presence of oxygen maximizes the energy output.
The process of aerobic respiration is generally represented using the following chemical equation. |
All living organisms, including both plants and animals, perform some kind of cellular respiration. Cellular respiration is a chemical pathway in which organic molecules, such as glucose, are broken down to release energy in a quickly usable form (e.g., ATP). The specific pathway is determined in part by the presence of oxygen.
These processes are shown in the diagram to the left. In general, one glucose molecule and six oxygen molecules are needed to produce six carbon dioxide molecules, six water molecules, and up to 38 ATP molecules. The actual number of ATP molecules varies because some of the energy may be used when substances are moved around the cell (e.g., from the cytoplasm to the mitochondria). This equation, however, is a summary of the overall process and does not represent all of the steps that occur during the process of aerobic respiration. In addition, organic molecules (other than glucose) can be processed and broken down as a source of energy to produce ATP molecules. These nutrients includes carbohydrates, lipids, and proteins. |
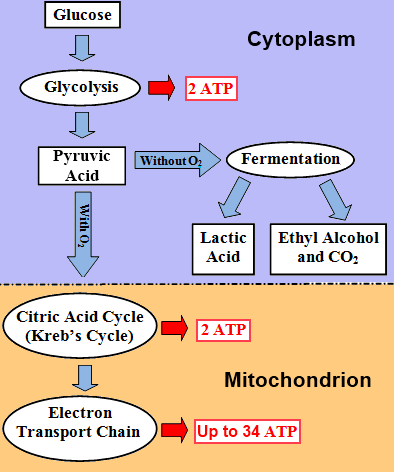
Glycolysis
Glycolysis is the first stage of cellular respiration, and it takes place in thecytoplasm. During glycolysis, a six-carbon glucose molecule is broken down with the aid of enzymes into two three-carbon pyruvic acid molecules.
Two ATP molecules are required to initiate the reaction, but four ATP molecules are produced. Therefore, glycolysis produces a net gain of two ATP.
A brief summary of glycolysis follows:
1 glucose molecule → 2 pyruvic acid molecules + 2 ATP molecules
Two ATP molecules are required to initiate the reaction, but four ATP molecules are produced. Therefore, glycolysis produces a net gain of two ATP.
A brief summary of glycolysis follows:
1 glucose molecule → 2 pyruvic acid molecules + 2 ATP molecules
Aerobic Respiration
Aerobic Respiration occurs in the presence of oxygen and is divided into two steps—the Krebs cycle (the citric acid cycle) and the electron transport chain.
- Citric Acid Cycle
The first step of aerobic respiration is called the citric acid or Krebs cycle.During this cycle, the pyruvic acid formed in glycolysis travels to themitochondria where it is chemically transformed in a series of steps, so carbon dioxide, water, and energy (2 ATP molecules) are eventually released. pyruvic acid → carbon dioxide + water + energy (2 ATP) - Electron Transport Chain
The second step of aerobic respiration is the electron transport chain. Most of the ATP molecules are formed during this part of the cycle. The electron transport chain is a series of chemical reactions ending with water, carbon dioxide, and a net yield of up to 34 ATP molecules. The ATP that is synthesized can be used by the cell for cellular metabolism.
Limiting Factors
Cellular respiration is driven by enzymes. Therefore, the reaction can only occur within a specific temperature and pH range. High temperatures and/or extreme pH levels cause the enzymes to denature. (When an enzyme denatures, it changes shape and becomes inactive.)
The availability of reactants affects the process of cellular respiration. In particular, oxygen is required for aerobic respiration to proceed normally.When oxygen is not present in sufficient quantities, cells use an anaerobic pathway of cellular respiration, which is far less efficient.
The availability of reactants affects the process of cellular respiration. In particular, oxygen is required for aerobic respiration to proceed normally.When oxygen is not present in sufficient quantities, cells use an anaerobic pathway of cellular respiration, which is far less efficient.
Anaerobic Respiration
If no oxygen is available, cells can obtain energy through the process of anaerobic respiration. A common anaerobic process is fermentation.Fermentation is not an efficient process and results in the formation of far fewer ATP molecules than aerobic respiration (i.e., fermentation may yield only 2 ATP, which is less than 10% of the amount of ATP produced by aerobic respiration).
There are two primary fermentation processes:
There are two primary fermentation processes:
- Lactic acid fermentation occurs when oxygen is not available (e.g., in muscle tissues during rapid and vigorous exercise when muscle cells may be depleted of oxygen). The pyruvic acid formed during glycolysis is broken down to lactic acid, and in the process, energy, which is used to form ATP, is released.
However, lactic acid is toxic to cells. As it builds up in the tissue, lactic acid causes a burning, painful sensation. So, this process is limited, and cells need oxygen for sustained activity. Lactic acid fermentation is summarized by the general equation:
- Alcohol fermentation occurs in yeasts and some bacteria. During this process, pyruvic acid formed during glycolysis is broken down to produce alcohol, carbon dioxide, and energy that is used to form ATP. The general equation for alcohol fermentation is summarized below: